Table of Contents
Cemented carbide, as a core material in the modern industrial system, possesses high hardness, wear resistance, and corrosion resistance, playing an irreplaceable role in the manufacturing of cutting tools, mold processing, the production of mining tools, and precision machining.
With the ever-increasing demand for cemented carbide in the industrial sector, global production of cemented carbide is growing by the day.
Generation and Resource Significance of Scrap Cemented Carbide
This growth trend has also led to the generation of large quantities of scrap cemented carbide.
These scrap materials contain abundant strategic scarce resources, with tungsten content ranging from 40% to 95% and cobalt content ranging from 3% to 20%.
Both tungsten and cobalt are classified as strategic scarce resources, and the scarcity of cobalt is particularly severe, with most cobalt resources relying on imports.
-
Importance of Recycling for Resource Security and Sustainability
Against this backdrop, scrap cemented carbide recycling serves as a critical step toward sustainable industrial development.
This process also holds profound significance for ensuring resource security and enhancing economic efficiency.
Cemented carbide cutting tools represent a widely used and typical product within the cemented carbide category, and the industry hails them as the “teeth” of modern manufacturing.
Industries extensively apply these tools in machining, automotive, and aerospace fields.
Their scrap volume accounts for the vast majority of cemented carbide waste and provides a stable and abundant source of raw materials for recycling.
In particular, cemented carbide cutting tools have high tungsten and cobalt content, relatively simple compositions, and few impurities, making them highly suitable for efficient, high-purity recycling and regeneration.
Scrap cutting tools are typically concentrated in machining workshops across various factories. The sources of these tools remain clearly identified.
This condition enables easy collection at low cost. The regular shapes of the tools facilitate pretreatment.
-
Overview of Recycling Technologies and High-Temperature Oxidation Method
Current recycling technologies for waste cemented carbide primarily include zinc fusion, mechanical crushing, electrochemical methods, acid leaching, and oxidation.
Each method has certain limitations; among the various recycling technologies, high-temperature oxidation is relatively more widely applied.
Due to its advantages—including a simple process, compact workflow, low energy consumption, and environmental friendliness—this method has become the preferred process in the field of cemented carbide recycling and reuse.
-
Research Focus and Objective
This paper focuses on coated cutting tools, a representative type of cemented carbide product, to conduct an in-depth investigation into the microscopic evolution of the subsurface oxide layer following high-temperature oxidation.
The study aims to provide theoretical guidance on the key parameters for achieving high efficiency and energy savings in cemented carbide oxidation recycling processes.
Test Conditions
Uncoated cemented carbide cutting tools serve as specimens for differential thermal analysis (DTA) and thermogravimetric analysis (TGA); Table 1 shows their matrix compositions.
A DTA-TGA instrument analyzes the oxidation process of the specimens.
The experiment holds the specimens at 523 °C, 723 °C, and 923 °C for 30 minutes each to determine the most suitable oxidation temperature and to record mass changes during oxidation.
A resistance furnace heats the test tools to 923 °C at a rate of 10 °C/min.
The process maintains the tools at 923 °C for 1, 2, 4, and 6 hours, respectively, to carry out oxidation in air.
| Element | WC | Co | TiC |
|---|---|---|---|
| Content (%) | 90 | 8 | 2 |
Table 1 Composition of Cemented Carbide Tool
Next, cemented carbide cutting tools with Al₂O₃ and TiCN composite coatings were placed in a resistance furnace.
The composition of these tools was identical to that of the uncoated tools.
The experiment heats the samples at the same rate to 923 °C and 1123 °C and maintains them at these temperatures for 2 hours. The process calculates the weight gain caused by oxidation after heating.
A JEOL JXA-8530F scanning electron microscope (SEM) observes the grain morphology of the subsurface oxide layer.
A Panalytical X-ray diffractometer (XRD) performs phase analysis on the oxidized coating products and the oxidized substrate products.
Oxidation Test Results and Analysis
-
Thermogravimetric and Differential Thermal Analysis
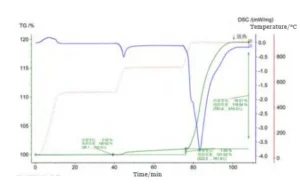
Figure 1 shows the thermogravimetric (TG) curves of uncoated cemented carbide at different temperatures.
As shown in Figure 1, at the initial temperature of 523 °C, the mass change curve shows no upward trend, indicating that no oxidation occurred at this temperature.
Subsequently, the curve for the cemented carbide rises slowly at 523 °C, indicating that the weight begins to increase gradually;
Starting at 781 °C, the rate of mass change accelerates sharply with rising temperature.
When held at 923 °C, the mass no longer increases, with the weight gain reaching 119.54% of the original mass.
The results indicate that cemented carbide begins oxidation at 523 °C. The oxidation rate reaches its maximum at 923 °C.
After this point, the oxidation rate gradually decreases until complete oxidation occurs.
At 923 °C, the process shows a rapid mass change. This stage produces a distinct exothermic peak. The result confirms the exothermic nature of the oxidation reaction.
-
Chemical Equation for the Oxidation Reaction
First, tungsten carbide begins to oxidize at temperatures above 523 °C, producing WO₃ and CO₂, which is consistent with the changes observed in thermogravimetric analysis.
This reaction is exothermic, and the chemical equation is:
2WC+5O2→2WO3+2CO2 (1)
When cobalt acts as the binder phase, the initial product of its reaction with oxygen is CoO, which is subsequently converted to Co₃O₄ in a secondary reaction. The reaction equations are as follows:
2Co+O2→2CoO (2)
3Co+2O2→Co3O4 (3)
It is known that the oxidation products of TiC include TiO₂ and CO₂, and the chemical equations are as follows:
TiC+2O2→TiO2+CO2 (4)
Finally, Co₃O₄ reacts further with WO₃ to form CoWO₄. The chemical equation is:
3WO3 + Co3O4 → 3CoWO4 + 1/2 O2 (5)
-
Effect of Oxidation Time on Oxidation Behavior
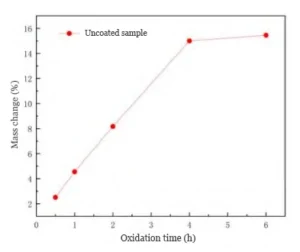
Figure 2 presents oxidation weight gain tests. The tests use uncoated cemented carbide cutting tools placed in flowing air at 923 °C.
The test results indicate that, under these temperature conditions, the oxidation weight gain process of the cemented carbide exhibits a distinct linear trend.
This paper evaluates oxidation behavior by defining the ratio of mass increase during the oxidation process as a measure of oxidation time effects. This ratio reflects the degree of oxidation.
Based on the TGA findings described earlier: at 923 °C, the cemented carbide is in a rapid oxidation phase.
As the oxidation time gradually increases, the mass gain due to oxidation increases at a constant rate; however, in the later stages of oxidation, the rate of mass gain slows down.
Oxidation continues for 6 hours and drives the cemented carbide to a fully oxidized state with no residual matrix remaining. The process records a final mass increase of 15.45%.
This value does not match the 19.54% mass increase observed in the TGA test for complete oxidation.
This indicates that some substances volatilize at high temperatures.
This is because, under high-temperature conditions, a portion of the generated WO₃ sublimated, resulting in an overall reduction in mass.
Even as the degree of oxidation continued to increase, oxygen was still able to come into contact with the matrix unimpeded.
At 923 °C, the oxidation process produces oxides on the surface of the cemented carbide tool.
These oxides fail to form a dense oxide layer. As a result, oxygen continues to contact the matrix without effective blockage.
-
Effect of Tool Coatings on Oxidation Behavior
When conducting high-temperature oxidation tests on coated tools, weighing measurements revealed that the mass remained essentially stable with no significant changes after 2 hours of continuous oxidation at a constant temperature of 923 °C.
The test temperature rises to 1123 °C, and oxidation continues for 2 hours. The tool mass increases significantly by 3.3% at this condition.
This result clearly indicates damage to the surface coating structure under high temperature.
Figure 3 shows SEM images of the subsurface of the coated tool after oxidation at 923 °C and 1123 °C.
Figure (a) shows the behavior under 923 °C oxidation conditions. The surface coating of the tool does not undergo oxidative decomposition.
Only the surface layer of alumina experiences partial decomposition. The underlying TiCN coating maintains an intact structure and remains well preserved.
Figure (b) presents a stark contrast, showing the surface of the material after oxidation at 1123 °C.
The surface coating undergoes destruction and decomposition. High-temperature conditions cause the coating components to decompose and volatilize.
The process exposes the substrate directly to a high-temperature oxygen environment. This exposure triggers oxidation of the substrate.
Formation and Characteristics of Subsurface Oxide Layers
Based on an in-depth analysis of the morphological characteristics of the subsurface oxide layers, these layers can be divided into two distinct stages.
Oxide layer 1, located at the outermost layer and in direct contact with oxygen, exhibits a loose, porous structure.
This structure makes the layer highly susceptible to peeling when subjected to external forces.
In contrast, subsurface oxidation layer 2, located near the substrate, forms a dense oxide structure.
This dense layer effectively blocks further contact between oxygen and the substrate, thereby inhibiting the continuation of subsequent oxidation reactions.
This phenomenon arises because, at a high temperature of 1123 °C, the WC substrate is rapidly oxidized, leading to the formation of a dense WO₃ oxide layer.
Figure 3(c) shows the microstructure of the oxide in region 1 of subsurface oxide layer 1.
The results indicate that the oxide formed in this region exhibits a granular distribution with numerous pores between the particles.
This structural characteristic makes the oxide in this region prone to dispersion when subjected to external forces.
Figure 3(d), on the other hand, presents the microstructure of region 2 in subsurface oxide layer 2, which is close to the matrix. Here, the tungsten oxide does not exhibit a granular morphology near the grains containing alloying additives.
This phenomenon indicates that the addition of alloying elements effectively inhibits the granular dispersion of tungsten oxide, promoting the formation of a denser oxide layer structure.
Consequently, this significantly increases the difficulty for oxygen to come into contact with the matrix and undergo subsequent oxidation reactions.

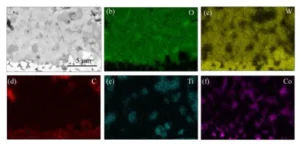
EDS Elemental Distribution in Subsurface Oxide Layers
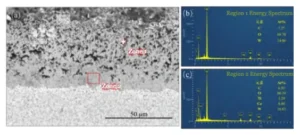
Figure 4 shows the results of energy-dispersive spectroscopy (EDS) elemental analysis performed on different subsurface oxide layers.
In Figure (b), the oxidized surface of the cemented carbide in subsurface oxide layer 1 consists primarily of W, O, and C.
Based on the “2.2 Chemical Equations for Oxidation Reactions” section above, the products should be WO₃ and trace amounts of residual WC;
Figure (c) shows Region 2 of the subsurface oxide layer 2 when compared with Region 1. The W content in Region 2 decreases significantly. Co and Ti elements also appear in this region.
The proportion of O remains unchanged. This result indicates that Ti and Co in Region 2 oxidize into corresponding oxide products.
Oxygen Penetration and Elemental Redistribution Mechanism
Figure 5 shows the EDS surface scan analysis of the subsurface oxide layer in Region 2.
Fig 5(b) shows that the O element from the oxidized surface fully penetrates the subsurface oxide layer and distributes uniformly throughout it.
The oxidation process reduces the concentrations of W and C in the oxide layer compared with the matrix because oxidation makes the WC matrix structure less dense.
Ti, in contrast, enriches in the dark solid solution and forms TiO₂ through oxygen pathways that the expansion and fracturing of WC create during oxidation.
The oxide structure of Ti is relatively dense and, together with WO₃, forms a dense oxide layer on the upper surface of the matrix.
At different temperatures, compared to the oxidation time of WC, the start and end times of Ti oxidation are later.
This implies that the presence of Ti confers greater stability and a slower oxidation rate to the internal solid solution, thereby hindering the subsequent entry of oxygen into the matrix to induce oxidation.
The concentration of Co elements in the subsurface is lower than that in the matrix interior, and its products are distributed non-uniformly on the surface of the dense oxide layer;
The distribution density of O content corresponding to the Co element distribution area is slightly higher than in other areas.
This is because Co begins to oxidize earlier than WC; under high-temperature oxidation at 1123 °C, Co rapidly diffuses, accelerating the oxidation of the cemented carbide and dispersing throughout the entire dense oxide layer within a short period of time.
Phase Composition of Oxidized Coating and Matrix
Figure 6 shows the XRD results obtained after oxidizing the coating and the substrate at 1123 °C.
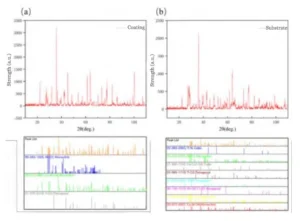
Figure 6 shows that the main products of the surface coating oxidation are WO₃, CoWO₄, and TiO₂, with calculated relative contents of 17%, 31%, and 51%, respectively; whereas the matrix interior consists of WO₃, CoWO₄, TiO₂, and Co₃O₄, with relative contents of 27%, 11%, 38%, and 17%, respectively.
It can be seen that after complete oxidation of the surface coating, TiO₂ accounts for the largest proportion at 51%, which is higher than the 38% found within the matrix.
This is because the presence of the surface TiCN coating causes the TiO₂ concentration to be slightly higher than that within the matrix.
Additionally, within the matrix, the oxidation of the Co phase occurs before that of WC; therefore, the Co elements in the matrix preferentially react with oxygen to form Co₃O₄.
Since this portion has not yet reacted with WO₃ to form CoWO₄, there is a difference in the distribution of Co₃O₄ between the matrix and the surface layer.
Conclusion
This study uses high-temperature oxidation to perform oxidation tests on cemented carbide cutting tools.
The process produces oxidized surfaces on both coated and uncoated cemented carbide cutting tools, and subsequent analysis systematically examines the microstructural morphology and phases of the subsurface layer after oxidation.
The main conclusions are as follows:
(1) Optimal Oxidation Temperature of Uncoated Cemented Carbide Tools
Thermal gravimetric analysis of oxidation at different temperatures determines the optimal oxidation temperature for uncoated cemented carbide tools as 923 °C.
At this temperature, the tools enter a rapid oxidation phase and achieve full oxidation.
(2) Oxidation Stages and Subsurface Grain Structure Evolution
Grain morphology and distribution characteristics of the subsurface after oxidation indicate the oxidation process.
The oxidation process divides into a complete oxidation stage and a densification stage. Distinct oxidation zones appear during the process.
In Oxidation Zone 1, the grain structure was loose, porous, and prone to flaking, whereas in Oxidation Zone 2, the grain structure formed a dense, cohesive oxide structure.
(3) Selective Oxidation Behavior of Ti Compared to WC
Compared to the oxidation time of WC, the onset and completion of Ti oxidation occur later.
This is because the presence of Ti confers greater stability and a slower oxidation rate to the solid solution, indicating that Ti exhibits selective oxidation behavior in cemented carbide.
(4) Phase Composition and Elemental Oxide Distribution
Phase analysis of the subsurface oxide layer reveals that the primary phases in the post-oxidation system are WO₃, CoWO₄, Co₃O₄, and TiO₂.
Migration and enrichment of TiO₂ components generated during the reaction within the surface coating primarily cause the high TiO₂ content at the surface.
Meanwhile, the presence of Co₃O₄ within the matrix is mainly due to the preferential oxidation of Co over WC, and the fact that some of the initially formed Co₃O₄ did not undergo a solid-state reaction with WO₃ to form CoWO₄.
FAQ
Impedit egestas aliquet?
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Sapien class quo temporibus?
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Elementum voluptate sodales?
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.




